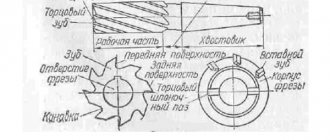
What happens to aluminum when heated?
It does not interact with concentrated sulfuric and hydrochloric acids at room temperature; when heated, it reacts to form salts, oxides and water.
Interesting materials:
How many hours a day should a one-year-old child sleep? How many people should be on the electrical safety commission? How many people should be subordinate to the boss? How many people should there be in management? How many dB should headphones have? How much money should be in the financial cushion? How many children should there be in a group per teacher? How many children should there be in a combined group? How many days should there be drainage after surgery? How many days should a vacation be?
Phenomena that occur in metal when heated
And heating modes
When heated, a layer of oxides is formed on the surface of the workpieces, called scale, the thickness of which depends on the temperature and heating time, the composition of the furnace atmosphere, the chemical composition of the alloy and the location of the workpieces in the furnace. Alloys oxidize most intensively at temperatures of 900...1200 °C.
Heating carbon steels also leads to burning out of the carbon of the surface layer to a depth of 2 mm. Reducing the carbon content, called decarburization, leads to a decrease in the strength and hardness of the steel. Decarburization is especially harmful for small-sized workpieces that have small allowances for machining and are subject to subsequent hardening.
To reduce scaling and decarburization, heating in a protective atmosphere or vacuum, high-speed heating, protective fills and coatings applied to the workpieces before heating are used.
High-carbon and high-alloy steels and many complex alloys with low thermal conductivity and ductility require slow heating to avoid cracks. Billets made of such steels and alloys are loaded into a furnace at a low temperature, maintained at this temperature to uniformly heat the workpiece throughout the entire volume, and then the temperature of the furnace is increased. When heated, large ingots made of alloy steel are subjected to several exposures at different temperatures.
The choice of heating mode before pressure treatment consists in determining a rational temperature range (temperatures of the beginning and end of treatment) and heating time. The lower limit of the temperature range of pressure treatment of steel billets exceeds 727 °C, and the upper limit should be 100...150 °C below the melting temperature. When heated to higher temperatures, two types of defects appear in the metal - overheating and burnout. When overheated, grain sizes increase, ductility decreases and mechanical properties deteriorate. This type of defect can be eliminated for some steels by additional pressure treatment and normalization.
Burnout is the oxidation of metal along grain boundaries when heated to temperatures close to the melting point. As a result, the bond between grains is disrupted and the metal is disrupted during processing. collapses under pressure. Overburning is an irreparable defect.
The processing temperature range depends on the grade of the alloy being processed. For steels, the start and end temperatures of pressure treatment can be determined from the Fe-C diagram (Fig. 16.9). The diagram shows that low-carbon steels have a wide (up to 500 °C) processing temperature range.
| Rice. 16.9.Fe-C state diagram: 1 - burnout; 2 - overheating; 3 - hot pressure treatment area |
The heating time is determined based on two conflicting requirements. On the one hand, in order to reduce the formation of scale and increase productivity, it is necessary to reduce the heating time by increasing its speed, on the other (in order to avoid the formation of cracks) it is necessary to reduce the heating rate and increase its duration. The latter is especially important for large-section workpieces made of high-alloy alloys. For carbon steel workpieces with a cross-section of up to 100 mm2, a high heating rate is allowed and they can be loaded cold into a furnace with a temperature of 1300 °C.
The heating time T (h) in this case can be determined by the formula N.N. Dobrokhotova
T = akD-jD,
where k is a coefficient depending on the steel grade (for carbon and low-alloy steels fe = 10, for high-carbon and high-alloy steels k - 20); D —diameter or side of the square of the workpiece, m; a is a coefficient that takes into account the method of laying workpieces in the oven (Fig. 16.10). The more densely the workpieces are laid in the furnace (the smaller the distance I), the greater the coefficient a and the longer the heating of the workpieces.
Billets made of high-alloy steels are heated in two stages due to their possible destruction as a result of the occurrence of thermal stresses at high heating rates:
| td |
| dJ3T ~amX |
| a = 1 |
sh)shsh>yat
a = 1.25
7SHSHSH7
Rice. 16.10. The influence of the method of laying blanks in the furnace on the coefficient a
First, they are slowly heated to 650 °C, and then, when the plasticity The total heating time is t-t1 + t2, where T and T2 are the times of the first and second heating stages, respectively:
tg
=al3,3Z)VD;
tz=o&,w4d.
cooling process (especially of workpieces made of alloy steels) during pressure treatment is a critical technological operation, which, if performed incorrectly, can lead to defects, since during cooling, cracks form in the workpieces more often than during heating. The cooling rate should not exceed permissible values. Billets made of low- and medium-carbon steels can be cooled in air individually or in groups on racks. Large forgings made of alloy steels are cooled slowly along with the furnace, allowing exposure for several hours at certain temperatures. The cooling cycle of billets depends on their chemical composition and size, and for large forgings it can last several days.
And heating modes
When heated, a layer of oxides is formed on the surface of the workpieces, called scale, the thickness of which depends on the temperature and heating time, the composition of the furnace atmosphere, the chemical composition of the alloy and the location of the workpieces in the furnace. Alloys oxidize most intensively at temperatures of 900...1200 °C.
Heating carbon steels also leads to burning out of the carbon of the surface layer to a depth of 2 mm. Reducing the carbon content, called decarburization, leads to a decrease in the strength and hardness of the steel. Decarburization is especially harmful for small-sized workpieces that have small allowances for machining and are subject to subsequent hardening.
To reduce scaling and decarburization, heating in a protective atmosphere or vacuum, high-speed heating, protective fills and coatings applied to the workpieces before heating are used.
High-carbon and high-alloy steels and many complex alloys with low thermal conductivity and ductility require slow heating to avoid cracks. Billets made of such steels and alloys are loaded into a furnace at a low temperature, maintained at this temperature to uniformly heat the workpiece throughout the entire volume, and then the temperature of the furnace is increased. When heated, large ingots made of alloy steel are subjected to several exposures at different temperatures.
The choice of heating mode before pressure treatment consists in determining a rational temperature range (temperatures of the beginning and end of treatment) and heating time. The lower limit of the temperature range of pressure treatment of steel billets exceeds 727 °C, and the upper limit should be 100...150 °C below the melting temperature. When heated to higher temperatures, two types of defects appear in the metal - overheating and burnout. When overheated, grain sizes increase, ductility decreases and mechanical properties deteriorate. This type of defect can be eliminated for some steels by additional pressure treatment and normalization.
Burnout is the oxidation of metal along grain boundaries when heated to temperatures close to the melting point. As a result, the bond between grains is disrupted and the metal is disrupted during processing. collapses under pressure. Overburning is an irreparable defect.
The processing temperature range depends on the grade of the alloy being processed. For steels, the start and end temperatures of pressure treatment can be determined from the Fe-C diagram (Fig. 16.9). The diagram shows that low-carbon steels have a wide (up to 500 °C) processing temperature range.
| Rice. 16.9.Fe-C state diagram: 1 - burnout; 2 - overheating; 3 - hot pressure treatment area |
The heating time is determined based on two conflicting requirements. On the one hand, in order to reduce the formation of scale and increase productivity, it is necessary to reduce the heating time by increasing its speed, on the other (in order to avoid the formation of cracks) it is necessary to reduce the heating rate and increase its duration. The latter is especially important for large-section workpieces made of high-alloy alloys. For carbon steel workpieces with a cross-section of up to 100 mm2, a high heating rate is allowed and they can be loaded cold into a furnace with a temperature of 1300 °C.
The heating time T (h) in this case can be determined by the formula N.N. Dobrokhotova
T = akD-jD,
where k is a coefficient depending on the steel grade (for carbon and low-alloy steels fe = 10, for high-carbon and high-alloy steels k - 20); D —diameter or side of the square of the workpiece, m; a is a coefficient that takes into account the method of laying workpieces in the oven (Fig. 16.10). The more densely the workpieces are laid in the furnace (the smaller the distance I), the greater the coefficient a and the longer the heating of the workpieces.
Billets made of high-alloy steels are heated in two stages due to their possible destruction as a result of the occurrence of thermal stresses at high heating rates:
| td |
| dJ3T ~amX |
| a = 1 |
sh)shsh>yat
a = 1.25
7SHSHSH7
Rice. 16.10. The influence of the method of laying blanks in the furnace on the coefficient a
First, they are slowly heated to 650 °C, and then, when the plasticity The total heating time is t-t1 + t2, where T and T2 are the times of the first and second heating stages, respectively:
tg
=al3,3Z)VD;
tz=o&,w4d.
cooling process (especially of workpieces made of alloy steels) during pressure treatment is a critical technological operation, which, if performed incorrectly, can lead to defects, since during cooling, cracks form in the workpieces more often than during heating. The cooling rate should not exceed permissible values. Billets made of low- and medium-carbon steels can be cooled in air individually or in groups on racks. Large forgings made of alloy steels are cooled slowly along with the furnace, allowing exposure for several hours at certain temperatures. The cooling cycle of billets depends on their chemical composition and size, and for large forgings it can last several days.
Temperature coefficient of linear expansion of metals, solids, liquids (Table)
The table shows the average values of the temperature coefficient of linear expansion ɑ of metals and alloys in the range from 0 to 100 °C (unless a different temperature is indicated).
| Metal, alloy | Linear expansion coefficient ɑ, 10-6°С-1 |
| Aluminum | 2,4 |
| Bronze | 13-21 |
| Tungsten (in the temperature range from 0 to 200 °C) | 4,5 |
| Duralumin (at t = 20 °C) | 23 |
| Gold | 14 |
| Iron | 12 |
| Invar* | 1,5 |
| Iridium | 6,5 |
| Constantan | 42339 |
| Brass | 17-19 |
| Manganin | 18 |
| Copper | 17 |
| Nickel silver | 18 |
| Nickel | 14 |
| Nichrome (from 20 to 100 °C) | 14 |
| Tin | 26 |
| Platinum | 9,1 |
| Platinite** (at t = 20 °C) | 41920 |
| Platinum-iridium*** (from 20 to 100 °C) | 8,8 |
| Lead | 29 |
| Silver | 20 |
| Carbon steel | 43009 |
| Zinc | 32 |
| Cast iron (from 20 to 100 °C). | 41952 |
| * This alloy has a very low linear thermal expansion coefficient. Used for the manufacture of parts for precision measuring instruments. ** Conductor material, the coefficient of linear expansion of which is the same as that of glass; used in the manufacture of electric lamps. *** The prototypes of the kilogram and meter were made from this alloy. |
Temperature coefficient of linear expansion of solids
The table shows the average values of the temperature coefficient of linear expansion ɑ of solids in the range from 0 to 100 °C (unless a different temperature is indicated).
| Substance | Linear expansion coefficient ɑ, 10-6°С-1 |
| Diamond | 1,2 |
| Concrete (at t = 20 °C) | 41913 |
| Granite (at t = 20 °C) | 8 |
| Graphite | 7,9 |
| Wood (at t = = 20 °C): | |
| - along the fibers | 5,5-5,5 |
| - across the fibers | 34-60 |
| Fused quartz (at * = 40 °C) | 0,4 |
| Brick (at t = 20 °C) | 41885 |
| Ice (in the temperature range from -20 to 0 °C) | 51 |
| Paraffin (from 16 to 48 °C) | 70* |
| Oak (from 2 to 34 °C): | |
| - along the fibers | 4,9 |
| - across the fibers | 54,4 |
| Pine (from 2 to 34 °C): | |
| - along the fibers | 5,4 |
| - across the fibers | 34 |
| Laboratory glass | 41885 |
| Window glass (from 20 to 200 °C) | 10 |
| Porcelain | 2,5-4,0 |
| Slate (at t = 20 °C) | 10 |
| * coefficient of volumetric expansion of paraffin. |