mining industry
Pyrometallurgy
This is a branch of mining metallurgy. It consists of the heat treatment of minerals, metallurgical ores and concentrates to effect physical and chemical transformations in the materials allowing the recovery of valuable metals.[1] Pyrometallurgical processing can produce marketable products such as pure metals, intermediates or alloys suitable as feed for further processing. Examples of elements recovered by pyrometallurgical processes include oxides of less reactive elements such as iron, copper, zinc, chromium, can, and manganese.[2]
Pyrometallurgical processes generally fall into one or more of the following categories:[ citation needed
]
- calcination,
- frying
- fuse
- cleaning
Most pyrometallurgical processes require an input of energy to maintain the temperature at which the process occurs. Energy is usually produced in the form of combustion or electrical heat. When sufficient material is present in the feed to maintain the process temperature solely by an exothermic reaction (i.e., without the addition of fuel or electrical heat), the process is called "autogenous". When processing some sulfide ores, the exothermic nature of their combustion is used.
Frying
Main article: Roasting (metallurgy)
Roasting consists of thermal gas-solid reactions that may include oxidation, reduction, chlorination, sulfation and pyrohydrolysis.
The most common example of roasting is the oxidation of metal sulfide ores. The metal sulfide is heated in the presence of air to a temperature that allows oxygen in the air to react with the sulfide to form sulfur dioxide gas and solid metal oxide. The solid product from roasting is often called " calcined"
".
In oxidative roasting, if the temperature and gas conditions are such that the sulfide feedstock is completely oxidized, the process is known as " dead roasting
".
Sometimes, as in the case of reverberatory or electric melting furnace feedstock pre-treatment, the firing process is carried out with less oxygen than required to completely oxidize the feedstock. In this case, the process is called " partial roasting
" because the sulfur is only partially removed.
Finally, if the temperature and gas conditions are controlled so that the sulfides in the feedstock react to form metal sulfates instead of metal oxides, the process is known as " sulfation roasting
".
Sometimes the temperature and gas conditions can be maintained such that the mixed sulfide feedstock (such as feedstock containing both copper sulfide and iron sulfide) reacts so that one metal forms the sulfate and the other forms the oxide, a process known as " spot roasting
" or “
selective sulfation
”.
General methods for obtaining metals
The significant chemical activity of metals (interaction with atmospheric oxygen, other non-metals, water, salt solutions, acids) leads to the fact that in the earth’s crust they are found mainly in the form of compounds: oxides, sulfides, sulfates, chlorides, carbonates, etc. In free form, there are metals located in the voltage series to the right of hydrogen (Ag, Hg, Pt, Au, Cu), although much more often copper and mercury can be found in nature in the form of compounds.
Minerals and ferrous rocks containing metals and their compounds, from which the isolation of pure metals is technically possible and economically feasible, are called ores
.
Obtaining metals from ores is the task of metallurgy.
Metallurgy
is both the science of industrial methods for obtaining metals from ores and a branch of industry.
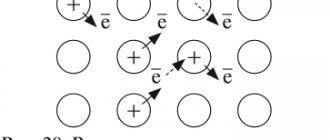
Any metallurgical process is a process of reduction of metal ions using various reducing agents. Its essence can be expressed as follows:
M n+ + ne−→M
To implement this process, it is necessary to take into account the activity of the metal, select a reducing agent, consider technological feasibility, economic and environmental factors.
In accordance with this, there are the following methods for obtaining metals:
• pyrometallurgical;
• hydrometallurgical;
• electrometallurgical.
Pyrometallurgy
Pyrometallurgy is the reduction of metals from ores at high temperatures using carbon, carbon monoxide (II), hydrogen, metals - aluminum, magnesium.
For example, tin is recovered from cassiterite SnO2, and copper from cuprite Cu2O
calcination with coal (coke):
SnO2+ 2С = Sn + 2СО ↑; Cu2O + C = 2Cu+ CO ↑
Sulfide ores are pre-roasted in the presence of air, and then the resulting oxide is reduced with coal:
2ZnS + 302 = 2ZnО + 2SO2 ↑; ZnO + C = Zn + CO ↑ sphalerite (zinc blende)
Metals are also isolated from carbonate ores by calcination with coal, since carbonates decompose when heated, turning into oxides, and the latter are reduced by coal:
FeСO3 = FeО + СO2 ↑ ; FeO + C = Fe + CO ↑ siderite (spar iron ore)
By reduction with coal one can obtain Fe, Cu, Zn, Cd, Ge, Sn, Pb and other metals that do not form strong carbides (compounds with carbon).
Hydrogen or active metals can be used as a reducing agent:
1) MoO3 + 3H2 = Mo + 3H2O (hydrothermy)
The advantages of this method include obtaining very pure metal.
2) TiO2+ 2Mg = Ti + 2MgO (magnesium thermia)
3МnO2 + 4Аl = 3Мn + 2Аl2O3 (aluminothermy)
Aluminum is most often used in metallothermy, the heat of oxide formation
which is very high (2A1 + 1.5 O2 = Al2O3 + 1676 kJ/mol). The electrochemical voltage series of metals cannot be used to determine the possibility of reactions occurring in the reduction of metals from their oxides. The possibility of this process can be approximately determined by calculating the thermal effect of the reaction (Q), knowing the values of the heats of formation of oxides:
Q= Σ Q1 - Σ Q 2,
Fuse
Main article: Smelting
Melting involves thermal reactions in which at least one product is a molten phase.
Metal oxides can be smelted by heating with coke or coal (a form of carbon), a reducing agent which releases oxygen in the form of carbon dioxide leaving the purified mineral. Concerns about carbon dioxide production are only a recent concern following the identification of the enhanced greenhouse effect.
Carbonate ores are also smelted using charcoal, but sometimes it is necessary to calcinate the former.[ citation needed
]
Other materials may be added as a stream, promoting the melting of oxide ores and promoting the formation of slag as the flux reacts with impurities such as silicon compounds. [ citation needed]
]
Smelting usually occurs at temperatures above the metal's melting point, but processes vary significantly depending on the ore used and other factors.[ citation needed
]
Pyrometallurgy
Pyrometallurgy
is a set of metallurgical processes occurring at high temperatures. This is a branch of metallurgy associated with the production and purification of metals and metal alloys at high temperatures, in contrast to hydrometallurgy, which involves low-temperature processes.
Description[ | ]
These are chemical processes occurring in metallurgical units at high (800-2000°C) temperatures. Therefore, pyrometallurgy is sometimes called “high temperature chemistry.”
Often chemical reactions are accompanied by a change in the aggregate state of the reacting substances: melting, sublimation, evaporation of the resulting metals or their compounds.
In such processes, interactions can occur between solid, liquid (melts) and gaseous phases in any combination.
Pyrometallurgical processes are the processes of agglomeration of metallurgical raw materials, smelting of charge materials, production of alloys, and refining of metals. In particular, this is roasting, blast furnace smelting, smelting in converters, arc and induction furnaces. Pyrometallurgy is the basis for the production of cast iron, steel, lead, copper, zinc, etc.
In pyrometallurgy, carbon reduction is often used - in cases where the metals being reduced do not form stable carbides, in addition to those mentioned above, such metals include germanium, cadmium, tin and others. In cases where stable carbides are formed by reduced metals, metallothermy is often used instead of reduction with carbon[1].
Pyrometallurgy is the main and most ancient field of metallurgy. From ancient times until the end of the 19th century, metal production was based almost exclusively on pyrometallurgical processes.
At the turn of the 19th and 20th centuries, another major branch of metallurgy, hydrometallurgy, acquired industrial importance.
However, pyrometallurgy continues to maintain a dominant position both in terms of production scale and variety of processes.
At the beginning of the 20th century, along with flame heating methods, different types of electric heating (arc, induction, etc.) began to be used in metallurgy; Around the same time, electrolysis of molten chemical compounds was introduced into industry (production of aluminum and other non-ferrous metals).
In the 2nd half of the 20th century, plasma melting of metals, zone melting, etc. became widespread. Metallurgical processes based on the use of electric current are classified as an independent field of pyrometallurgy - electrometallurgy.
Basic processes[ | ]
The main process of pyrometallurgy is ore smelting, which is carried out at such high temperatures that the products of chemical interaction melt, forming two liquid phases - metal or sulfide and slag. There are reduction and oxidation melts.
The defining process of reduction smelting is the reduction of metal oxides to ultimately produce a molten metal or its alloy with other elements. A typical reduction smelting process is the production of pig iron in blast furnaces. Reduction processes are also the main ones in the smelting of manganese, oxidized nickel, lead, and titanium ores.
The main reducing agents are carbon, carbon monoxide and hydrogen. Carbon monoxide is formed in the furnace itself when carbon is not completely burned; The main amount of hydrogen is obtained as a result of the decomposition of natural gas blown into the furnace.
A type of reduction smelting is the metallothermic production of metals, in which another metal with a greater affinity for oxygen is used as a reducing agent for a certain metal (Mn, Cr, V, etc.): Ca; Mg; Al and also Si. One of the advantages of metallothermic reduction is the production of metals that are not contaminated by carbon or hydrogen.
A typical oxidative ore smelting process is the processing of rich copper sulfide ores in shaft furnaces. During smelting, the bulk of the sulfur in sulfide minerals is oxidized, resulting in the release of a significant amount of heat. The main target product of smelting is the melt of FeS and Cu2S sulfides - matte.
Pig iron and ore smelting matte are essentially intermediate products that require additional processing. This treatment involves blowing the melts with air or pure oxygen, as a result of which the impurities contained in the alloys are oxidized and pass either into slag (SiO2; MnO; FeO, etc.) or into gas (CO; SO2). The process is called conversion.
A similar process to conversion is the fumming process—blowing slag melts with gas. Its difference from converting is that the metal melt is blown with an oxidizing gas, and when fumigating the slag, with a reducing gas.
And secondly, the products of oxidation of the metal melt - metal oxides - form a second liquid phase - slag, and the products of slag fuming - reduced volatile metals (or sulfides) in a vapor state are removed from the reaction space by a gas flow [2].
Literature[ | ]
Hydrometallurgy
Hydrometallurgy is the reduction of metals from their salts in solution.
The process takes place in two stages: 1) the natural compound is dissolved in a suitable reagent to obtain a solution of a salt of this metal; 2) from the resulting solution, this metal is displaced by a more active one or reduced by electrolysis. For example, to obtain copper from ore containing copper oxide CuO, it is treated with dilute sulfuric acid:
CuO + H2SO4 = CuSO4 + H2
Copper is then either removed from the salt solution by electrolysis or displaced from the sulfate by iron:
СuSO4. + Fe = Cu + FeSO4
In this way, silver, zinc, molybdenum, gold, and uranium are obtained.
Types of alloys
Despite such a variety of alloys, those based on iron and aluminum are of greatest importance to people. They are the ones that are most often encountered in everyday life. There are different types of alloys. Moreover, they are divided according to several criteria. Thus, various methods of producing alloys are used. According to this criterion they are divided into:
• Cast, which are obtained by melt crystallization of mixed components.
• Powder, created by pressing a mixture of powders and subsequent sintering at high temperature. Moreover, often the components of such alloys are not only simple chemical elements, but also their various compounds, such as titanium or tungsten carbides in hard alloys. Their addition in certain quantities changes the properties of metallic materials.
Methods for producing alloys in the form of a finished product or billet are divided into:
• foundries (silumin, cast iron);
• deformable (steel);
• powder (titanium, tungsten).
Properties of ores
Answering the question: what properties does iron ore have is not entirely simple. If only because the list of properties depends on the percentage of a given metal in the ore and the amount of foreign impurities. For example, red iron ore containing hematite (Fe2O3) contains as much as 70% of the total iron.
In general, by the way, only those where the ores contain 40% iron or more are considered viable iron mining. This figure really makes it clear that iron is widespread in the surrounding world many times more than other elements. For example, for the same uranium, its content in the ore in the amount of 2% would be considered an unprecedented success...
But let's return to our red iron ore. When characterizing iron ore, we can say that red iron ore ranges from a powdery substance to a dense one.
Limonite (also known as brown iron ore) is also an iron ore, but it is a porous and friable rock containing significant proportions of phosphorus and manganese. Clay is often used as waste rock. Due to this, by the way, it is quite easy to extract iron. That’s why cast iron is often made from it.
Production and consumption of metals
Distribution and areas of application
Of the most valuable and important metals for modern technology, only a few are found in large quantities in the earth’s crust: aluminum (8.9%), iron (4.65%), magnesium (2.1%), titanium (0.63%) . The natural resources of some very important metals are measured in hundredths and even thousandths of a percent. Nature is especially poor in noble and rare metals.
The production and consumption of metals in the world is constantly growing. Over the past 20 years, the annual global consumption of metals and the global metal stock have doubled and amount to about 800 million tons and about 8 billion tons, respectively. The share of products manufactured using ferrous and non-ferrous metals currently accounts for 72-74% of the gross national product of states. Metals in the 21st century remain the main structural materials, since their properties, economics of production and consumption are unmatched in most areas of application.
Of the 800 million tons of metals consumed annually, more than 90% (750 million tons) is steel, about 3% (20-22 million tons) is aluminum, 1.5% (8-10 million tons) is copper, 5-6 million t - zinc, 4-5 million tons - lead (the rest - less than 1 million tons). The scale of production of non-ferrous metals such as aluminum, copper, zinc, lead is measured in million tons/year; such as magnesium, titanium, nickel, cobalt, molybdenum, tungsten - in thousand tons, such as selenium, tellurium, gold, platinum - in tons, such as iridium, osmium, etc. - in kilograms.
Currently, the bulk of metals are produced and consumed in countries such as the USA, Japan, China, Russia, Germany, Ukraine, France, Italy, Great Britain and others.
Due to their physical properties (hardness, high density, melting point, electrical conductivity, sound conductivity, appearance and others), they are used in various fields. The use of metals depends on their individual properties:
- Iron and steel are hard and durable. Due to these properties, they are widely used in construction.
- Aluminum is forged, conducts heat well, and has high strength at ultra-low temperatures. It is used to make pans and foil in the cryogenic technique. Due to its low density - in the manufacture of aircraft parts.
- Copper has ductility and high electrical conductivity. That is why it has found its wide application in the production of electrical cables and power engineering.
- Gold and silver are very viscous, viscous and inert, have a high value, and are used in jewelry. Gold is also used to make non-oxidizing electrical connections.
Alloys and their applications
In their pure form, metals are rarely used. Metal alloys are much more widely used, as they have special individual properties. The most commonly used alloys are aluminum, chromium, copper, iron, magnesium, nickel, titanium and zinc. Much effort has been devoted to the study of iron and carbon alloys. Regular carbon steel is used to create low-cost, high-strength products when weight and corrosion are not critical.
Stainless or galvanized steel is used when corrosion resistance is important. Aluminum and magnesium alloys are used when strength and lightness are required. Copper-nickel alloys (such as monel metal) are used in corrosive environments and for the manufacture of non-magnetizable products
Nickel-based superalloys (eg Inconel) are used in high temperature applications (turbochargers, heat exchangers, etc.). At very high temperatures, single-crystal alloys are used.
Copper-nickel alloys (such as Monel metal) are used in corrosive environments and for the manufacture of non-magnetizable products. Nickel-based superalloys (eg Inconel) are used in high temperature applications (turbochargers, heat exchangers, etc.). At very high temperatures, single-crystal alloys are used.
Equipment
Various equipment is used for production and processing:
- For heat treatment - furnaces, smelters, forges.
- To change the roughness of surfaces - grinding machines, sandblasting.
- To create recesses, process edges, ends - slotting, drilling, milling machines.
- To give a simple or complex cylindrical shape - lathes.
- For cutting workpieces - saws, laser or waterjet cutters.
Modern equipment is equipped with automatic control systems, which speeds up production and minimizes physical costs on the part of humans.
Homemade horn (Photo: Instagram / vetal7070)
Copper production in Russia and the world
According to analytical agencies, the Russian Federation confidently occupies fifth position among countries engaged in the mining and production of pure copper. Copper production in Russia on average per year is 860 thousand tons. The basis of the modern structure of copper production is made up of three large holdings: OJSC MMC Norilsk Nickel (Norilsk Nickel), LLC UGMKHolding (UMMC) and CJSC Russian Copper Company (RMK). These companies carry out a full production cycle from ore mining to the production of finished ingots, rolled products and wire. Each holding includes several enterprises equipped with the most advanced production technologies. Thanks to dynamic development, last year it was possible to increase copper production by seven percent.
Global copper production is fairly consolidated. Almost 35% of this metal is produced by the five largest companies. These include:
- Codelco (Chile).
- Freeport-McMoRan (USA).
- Glencore (Switzerland).
- BHP Billiton (Australia).
- Southern Copper (Mexico).
These companies obtain almost 80% of copper from primary raw materials (that is, they carry out a full processing cycle) and produce 20% as a result of processing incoming scrap. In Europe, the largest copper producers are: Poland, Portugal and Bulgaria. Each plant is capable of producing a wide range of copper products. Despite the current crisis, copper still remains a sought-after metal. One of the serious disadvantages inherent in this production is environmental problems. Assessments of emissions from copper smelters showed high levels of ambient air pollution. It contains a large number of chemical compounds harmful to health (cadmium, mercury, arsenic, lead, nitrogen oxides and carbon).
Features of copper ores
Copper-containing ores are characterized as multi-element. The most common connections are with:
- iron;
- gray;
- copper.
The following may be present in minor concentrations:
- nickel;
- gold;
- platinum;
- silver.
Deposits all over the world have approximately the same set of chemical elements in the ore composition; they differ only in their percentages. To obtain pure metal, various industrial methods are used. Almost 90% of metallurgical enterprises use the same method for producing pure copper - pyrometallurgical.
One of the largest ore mines produces 17 million tons of copper per year
The design of this process also makes it possible to obtain metal from recycled materials, which is a significant advantage for industry. Since the deposits belong to the group of non-renewable deposits, reserves decrease every year, ores become poorer, and their extraction and production becomes expensive. This ultimately affects the price of the metal on the international market. In addition to the pyrometallurgical method, there are other methods:
- hydrometallurgical;
- fire refining method.