| Corrosion is the process of destruction of metals and metal structures under the influence of various environmental factors - oxygen, moisture, harmful impurities in the air. |
The corrosion resistance of a metal depends on its nature, the nature of the environment and temperature.
- Noble metals do not corrode due to chemical inertness.
- Metals Al, Ti, Zn, Cr, Ni have dense gas-tight oxide films that prevent corrosion.
- Metals with a loose oxide film – Fe, Cu and others – are corrosion unstable. Iron is especially susceptible to rust.
There are chemical and electrochemical corrosion.
| Chemical corrosion is accompanied by chemical reactions. As a rule, chemical corrosion of metals occurs when the metal is exposed to dry gases; it is also called gas corrosion. |
3Fe + 2O2 = Fe3O4
With chemical corrosion, the following processes are also possible:
Fe + 2HCl → FeCl2 + H2
2Fe + 3Cl2 → 2FeCl3
As a rule, such processes take place in chemical production equipment.
| Electrochemical corrosion is a process of metal destruction, which is accompanied by electrochemical processes. As a rule, electrochemical corrosion occurs in the presence of water and oxygen, or in electrolyte solutions. |
In such solutions, processes of electron transfer from the metal to the oxidizing agent, which is either oxygen or an acid contained in the solution, occur on the surface of the metal.
In this case, the electrodes are the metal itself (for example, iron) and the impurities it contains (usually less active metals, for example, tin).
In such a contaminated metal, electrons are transferred from iron to tin, while the iron (anode) dissolves, i.e. is subject to corrosion:
Fe –2e = Fe 2+
On the surface of the tin (cathode), the process of hydrogen reduction from water or dissolved oxygen takes place:
2H+ + 2e → H2
O2 + 2H2O + 4e → 4OH–
| For example , when iron comes into contact with tin in a solution of hydrochloric acid, the following processes occur: |
Anode : Fe –2e → Fe 2+
Cathode : 2H+ + 2e → H2
Total reaction: Fe + 2H+ → H2 + Fe2+
If the reaction takes place under atmospheric conditions in water , oxygen participates in it and the following processes occur:
Anode : Fe –2e → Fe 2+
Cathode: O2 + 2H2O + 4e → 4OH–
Total reaction:
Fe 2+ + 2 OH – → Fe ( OH )2
4 Fe ( OH )2 + O 2 + 2 H 2 O → 4 Fe ( OH )3
This causes rust to form.
Causes
Causes of corrosion processes:
- contact of different types of metals, alloys;
- frequent temperature changes;
- friction between metal surfaces;
- prolonged exposure to moisture;
- influence of acids, alkalis, chemical elements;
- use of low-quality liquids during mechanical processing of materials;
- grease stains remaining on metal surfaces after touching them.
Rust can form from periodic exposure to static or direct current.
Classification of corrosion processes according to the conditions under which corrosion occurs.
- Gas corrosion occurs in the gas phase with a minimal amount of moisture. This corrosion occurs when metals come into contact with aggressive gases (halogens, oxygen, sulfur oxide).
- Atmospheric corrosion occurs in an atmosphere of air or other moist gas.
- Liquid corrosion is corrosion that occurs in various types of liquids.
- Underground corrosion is metal corrosion that occurs as a result of inhomogeneities in the soil.
- Corrosion under cryptoclimate conditions occurs in confined spaces.
- Radiation corrosion is caused by exposure to radiation.
- Marine corrosion occurs due to the depassivating properties of chlorine ions.
- Structural corrosion is associated with the structural heterogeneity of metals.
- Corrosion caused by stray currents.
Kinds
Corrosion processes are classified depending on different criteria. The main ones are color, mechanism of rust formation, type of aggressive environment, nature of destruction.
By color
Depending on the color, there are different types of rust. It can be black, yellow, brown, red. The shade depends on the chemical formula of the resulting substance.
Rusty metal
Yellow
The chemical formula of yellow rust is FeO(OH)H2O. It appears under the influence of high humidity, in an environment with a small amount of oxygen. This type of rust can be seen underwater.
Brown
The chemical formula of brown rust is Fe2O3. It is extremely rare and appears without exposure to moisture.
Red
The chemical formula of red rust is Fe2O3•H2O. Formed by simultaneous exposure to water and oxygen. It is more common than other species. The destructive process proceeds evenly, gradually spreading over the entire surface.
Black
Chemical formula: Fe3O4. Appears without exposure to moisture, in an environment with a small amount of oxygen. Often used to create superconductors because it is ferromagnetic.
According to the flow mechanism
Kinds:
- chemical;
- electromechanical.
The processes differ in the mechanism of material destruction.
Chemical
The process of metal destruction, provoking the disintegration of metal bonds, the development of chemical reactions between the atoms of the material. Elements that interact with each other are not spatially separated. The rate of destruction of a part depends on the speed of the chemical reaction.
Electrochemical
This process of destruction of metal parts occurs in an electrolyte environment and is combined with the generation of current.
Rusty ship
By type of aggressive environment
Kinds:
- Atmospheric.
- Gas.
- Radiation.
- Underground.
- Contact.
- Biocorrosion.
- Electrical corrosion.
- Corrosive cavitation.
- Stress corrosion.
- Fretting corrosion.
Atmospheric
Natural process of destruction. Can occur in air or gas atmosphere. An important condition is a high level of humidity. The higher it is, the faster the material will collapse.
Gas
The process of destruction of metal parts, which occurs in a gas environment. Features low humidity levels. The process of rust formation accelerates as the temperature rises.
Radiation
Occurs under intense exposure to radiation. In high-density alloys it occurs slowly.
Underground
If a metal part lies underground for some time, you may notice a green coating or other color distortions on its surfaces. This is a consequence of oxidative processes that occur in different types of soil.
Contact
Appears quickly in places where two different metals come into contact with each other. This is due to the difference in stationary potential in the electrolyte.
Biocorrosion
The process of destruction of metal parts, which is caused by the influence of various microorganisms and their metabolic products.
Rusty shipwrecks
Corrosion by electric shock
Can occur when exposed to stray or external current. The rate at which rust spreads depends on the current strength, duration, and frequency of its impact on metal parts.
Corrosive cavitation
One of the many processes of self-destruction of different types of metals. It starts when exposed to the external environment, mechanical damage.
Stress Corrosion
The process of destruction of alloys, which occurs when mechanical stress interacts with a corrosive environment. This type of corrosion is dangerous for metal structures that are subject to heavy loads.
Fretting corrosion
A complex corrosion process that occurs under the influence of a corrosive environment with various vibrations. To prevent the formation of rust, it is important to reduce the coefficient of friction of metal parts.
By nature of destruction
Kinds:
- solid;
- selective;
- local;
- subsurface;
- intercrystalline;
- slotted.
They differ in location, degree of penetration into the material, and severity of destruction.
Solid
With this corrosion process, all metal surfaces become covered with rust. It can be uniform or uneven, depending on the rate of destruction of the material in different places of the part.
Electoral
A similar process affects one of the elements of the metal structure, which does not have an anti-corrosion coating that slows down the destruction process.
Rusty car (Photo: pixabay.com)
Local
Rust spots are scattered on the metal surface. They are recesses of different sizes, some of which may be superficial, others through.
Subsurface
Appears under metal surfaces. It quickly penetrates deep into the material. This type of corrosion process is characterized by metal delamination.
Intercrystalline
Begins to appear along the boundaries of individual grains of the material. It is extremely difficult to identify by appearance. Indicators of density, strength, and ductility quickly deteriorate. Parts become fragile.
Slotted
Formed at the junction of two metal parts. May appear in technological gaps, under technical gaskets.
Classification of corrosion processes according to the determining mechanism.
2.1 Chemical corrosion of metals.
Chemical corrosion
is the process of interaction of metal with a corrosive environment. Such corrosion is not accompanied by the flow of electric current and usually occurs in the absence of moisture.
Chemical corrosion obeys the laws of kinetics of heterogeneous reactions. An example is the oxidation of magnesium in air:
2Mg + O2 = 2MgO
The thermodynamic stability of a metal can be assessed by the change in the Gibbs free energy in the corrosion reaction. Corrosion is possible if the Gibbs energy is less than zero ∆G <0.
For example, for the oxidation of iron in air according to the reaction:
3Fe + 2O2 = FeO + Fe2O3
the change in Gibbs energy has the form:
∆G =υ∆G0FeO + υ∆G0Fe2O3 – υ∆G0Fe – υ∆G0O2
Where:
∆Gx0 – change in the Gibbs energy of individual components, kJ/mol; υ – stoichiometric reaction coefficient.
∆G0 = 1*(-244.3) +1*(-740.3) – 3*0 – 2*0 = -984.6 kJ/mol. ∆G0 = -984.6 kJ/mol, from which we can conclude that the iron oxidation reaction proceeds spontaneously.
Examples of changes in ∆G for some other metals are presented in Table 1.
Table 1 - Change in Gibbs energy during ionization of metals.
| Reaction | ∆G, kJ/mol | Reaction | ∆G, kJ/mol |
| 4Na+O2 = 2Na2O | -340,4 | 4Cr+3O2 = 2Cr2O3 | -15,3 |
| 2Mg+O2 = 2MgO | -307,4 | 2Ni+ O2 =2NiO | -102,6 |
| 4Al+3O2 = 2Al2O3 | -239,4 | 2Pb+ O2 =2PbO | -90,9 |
| 2Zn+O2 = 2ZnO | -153,6 | 4Ag + O2 =2Ag2O | -8,51 |
| 4Au+3O2 = 2Au2O3 | +65,73 | 2Pt + O2 =2PtO | +36 |
Depending on operating conditions, the stability of metals can vary significantly. For most of them, the thermodynamic probability of oxidation decreases with increasing temperature.
2.2 Electrochemical corrosion of metals.
Electrochemical corrosion
– this is the process of interaction of a metal with a corrosive environment (electrolyte), accompanied by oxidation of the metal and restoration of the acceptor from the corrosive environment.
Electrochemical corrosion obeys the laws of electrochemical kinetics. It represents the transition of a metal from a free state to one associated with the loss of electrons. The rate of such corrosion depends on the electrode potential of the metal itself.
The modern theory explaining electrochemical corrosion was proposed by the Soviet scientist, Doctor of Chemical Sciences Yakov Mikhailovich Kolotyrkin.
This theory is based on the assumption that the entire surface of the metal is equally accessible to both the cathodic and anodic components of the corrosion process.
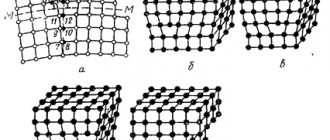
Electrochemical corrosion includes 4 main stages (Figure 2):
- Dissolution of metal Me (ionization) and release of electrons (e);
- Transfer of electrons through the metal (their number in the elementary stage - z);
- Restoring depolarizer D (attachment e);
- Transport of ions through an electrolyte solution.
Figure 2 – Scheme of operation of the corrosion element.
Electrochemical corrosion is a combination of two reactions occurring:
• at the anode:
Me0 - ze = MeZ+
• at the cathode:
DZ+ + ze = D0
The thermodynamic possibility of electrochemical corrosion is determined by the equilibrium potential; if it is greater than 0, then corrosion is possible.
∆Ep = Eox - Ered = ED - EMe> 0
From the formula we can conclude that the reaction is possible if and only if the depolarizer potential is greater than the metal potential ED > EMe. Depolarizers in aqueous solutions:
• in acidic environments:
2H+ + 2e = H2 O2 + 4H+ + 4e = 2H2O
• in neutral and alkaline:
2H2O + 2e = H2 + 2OH- O2 + 2H2O + 4e = 4OH-
If a hydroxyl ion or water acts as a depolarizer, then it is customary to say that corrosion occurs with hydrogen depolarization; if oxygen acts as a depolarizer, oxygen depolarization occurs. Very often there are cases of mixed depolarization, in which both hydrogen and oxygen are simultaneously reduced.
2.3 Biological corrosion of metals.
Biocorrosion
– this is the destruction of metal caused directly or indirectly by the activity of bacteria, mold and fungi.
Absolutely all materials and products known to man are susceptible to this type of corrosion. This is due to the formation of oxidative enzymes that cause the destruction of various substrates with the formation of organic acids. These acids also have a destructive effect on materials.
For example, organic acids produced by fungi cause pitting corrosion of carbon steel in holds or aluminum fuel tanks.
2.4 Cavitation corrosion of metals.
Cavitation corrosion
- this is destruction caused by an impact under the influence of a fluid medium (liquid or gas flow). It appears when gas bubbles collapse on the metal surface, accompanied by hydraulic shock.
2.5 Erosion.
Erosion
– wear and knocking out of metal particles from the surface under the influence of solid particles in the fluid flow. Metal erosion is accelerated by the corrosive effect of the environment. In other words, the loss of material occurs not only due to the mechanical action of the flow, but also as a result of some electrochemical process. An illustration of the erosion process is shown in Figure 3.
Figure 3 – Example of erosive corrosion.
Protection methods
To protect metal surfaces from corrosion, various techniques are used. Each of them is unique and has certain characteristics.
Application of protective coating
Protective coatings can be of two types - metallic and non-metallic. Types of non-metallic coatings:
- Chemical layer. More often these are oxide films that form on the surface under the influence of steam and air. One of the oxidation options is to immerse parts in a solution of nitric acid heated to 140°C.
- Paint and varnish coatings. The main disadvantage of paint and varnish coatings is their low resistance to temperature changes and mechanical damage.
- Powder paints. They are applied using specialized equipment in closed painting booths.
- Various polymer coatings.
Corrosion of individual metals.
The annual corrosion rates of some metals and alloys in various environments are given in Table 2. Table 2 - Corrosion rates mm/year of structural metals in various environments.
| Metal | Sulfur 5% | Acetic 5% | Nitrogen 5% | NaOH 5% | Fresh water | Sea water | City air |
| Aluminum | 8-100 | 0,5-5 | 15-80 | 13000 | 0,1 | 1-50 | 0-0,5 |
| Zinc-tin | High | 600-800 | High | 0,5-10 | 0,5-10 | 0,5-10* | 0-0,2 |
| Lead- iron | 2-500- | 2-500* | 100-1000 | 0-0,5 | 0-0,5 | 0,2-15 | 0-0,2 |
| Silicon- iron | 0-2 | 10-150* | 100-6000 | 0,1-2 | 0,1-2 | 0,2-15 | 0-0,2 |
| Steel | 15-100* | 10-100 | 1000-10000 | 0,1-10* | 0,1-10* | 0,1-10* | 0,2 |
| Stainless steel | 0-5 | 0-0,2 | 20 | 0-0,2 | 0-0,2 | 0-3 | 0-0,2 |
| Copper alloys | 0-100 | 0-0,5 | 0-2 | 0-0,2 | 0-0,2 | 0-200 | <0,1 |
| Nickel | 2-50 | 2-15* | 150-1500 | 0-1 | 0-1 | 0,2-15* | <0,1 |
| Nickel alloys | 2-35* | 2-10* | 0,1-1500 | 0-1 | 0-1 | 0,2-15* | <0,1 |
| Titanium | 10-100 | <0,1 | <0,1-1 | <0,2 | <0,1 | <0,1 | <0,1 |
| Molybdenum | 0-0,2 | <0,1 | High | <0,1 | <0,1 | <0,1 | <0,1 |
| Zirconium | <0,5 | <0,1 | <0,1 | <0,1 | <0,1 | <0,1 | <0,1 |
| Tantalum | <0,1 | <0,1 | High | <0,1 | <0,1 | <0,1 | <0,1 |
| Silver | 0-1 | <0,1 | <0,1 | <0,1 | <0,1 | <0,1 | — |
| Platinum | <0,1 | <0,1 | <0,1 | <0,1 | <0,1 | <0,1 | — |
| Gold | <0,1 | <0,1 | <0,1 | <0,1 | <0,1 | <0,1 | — |
5.1 Corrosion of iron and steels.
The most common structural metals were and remain iron-based alloys - steel (C < 2.14% of the total mass) and cast iron (C > 2.14% of the total mass). Iron is the metal most susceptible to atmospheric corrosion due to the release of hydrogen and oxygen depolarization. For steel, pitting corrosion is the most dangerous.
Corrosion of iron begins to occur at relative humidity above 75%. If the oxygen necessary for depolarization is absent, then iron (III) ions can act as an electron acceptor, being reduced to iron (II), which then react with oxygen and are oxidized again to iron (III).
2Fe2+ + 2H+ + 0.5O2 = 2Fe3+ + H2O
Thus, iron (III) ions act as an oxygen carrier, accelerating corrosion. The corrosion reaction of iron in atmospheric air is given at the beginning of the article.
The formation of an oxide protective film on the surface of iron is possible only in the presence of a strong oxidizing agent, for example, nitric acid. Under normal conditions, the oxide film on iron is loose and porous, i.e. unable to protect it from corrosion.
To protect against corrosion, steel is alloyed by adding certain components. Their influence can be summarized as follows:
- Copper up to 0.2% increases corrosion resistance in atmospheric conditions.
- Phosphorus up to 0.1% in combination with copper also increases corrosion resistance, but does not protect against pitting.
- Chromium and nickel reduce the rate of corrosion due to the fact that they form a protective film on the surface of the steel. Chromium in an amount of 13% significantly slows down the corrosion rate. When alloyed with more than 30% chromium, the steel also becomes resistant to pitting corrosion.
- Silicon increases resistance to pitting corrosion, but increases the likelihood of intergranular corrosion.
When using iron-based materials in coastal atmospheres, cathodic protection should be used. In industry, the surface of steels should be protected from corrosion by protective coatings and suitable corrosion inhibitors should be used. Most often, zinc and its alloys, cadmium, powder-polymer and zinc-rich paints are used as a protective coating.
5.2 Corrosion of aluminum and its alloys.
The second most popular structural material is aluminum-based alloys. The potential of aluminum is -1.66 V, which indicates its high activity and reactivity. Despite this, aluminum has a greater tendency to passivation and is highly resistant to atmospheric corrosion. In the passive state, the surface of aluminum is covered with a thin film of oxides (bayerite). However, this film is only stable in neutral environments (4.5 < pH < 8.5). In concentrated non-oxidizing acids or alkalis, aluminum products are subject to severe corrosion.
The reaction of aluminum with oxygen:
4Al + 3O2 = 2Al2O3
Reaction of aluminum with water:
2Al + 6H2O = 2Al(OH)3 + 3H2
Pure aluminum exhibits high corrosion resistance and ductility, but has low mechanical strength. Alloying components are added to increase strength and heat treatment ability.
The addition of alloying components, the potential of which is more electropositive than the potential of aluminum, leads to the formation of cathode and anodic zones. For example, when copper is added to an alloy, the intermetallic phase CuAl3 is formed. Being a cathode in relation to the base metal, CuAl3 is deposited at the grain boundaries, due to which the areas near these boundaries are depleted in copper and will act as anodes in relation to the grain boundaries themselves, causing intergranular corrosion.
A special case of aluminum corrosion involving intermetallic compounds is the so-called “black spots”. This type of corrosion occurs in three stages:
- the appearance of black dots on the surface of aluminum in areas of local concentration of intermetallic compounds;
- clear manifestation of the boundaries of intermetallic compounds,
- dissolution of intermetallic compounds and formation of discontinuities in the form of deep pittings and ulcers.
This effect is especially pronounced on large parts. Dots can often occur when etching and anodizing aluminum. Immediately after etching, the dots may not be visible, because they are hidden under a layer of pickling sludge. But if the part is washed and allowed to air dry, then over time the sludge turns yellow and black dots become easily visible to the naked eye (Figure 8).
Figure 8 - Example of black dots on etched aluminum D16.
Standard electrode potentials of various aluminum alloys and some intermetallic compounds are given in Table 3.
Table 3 - Standard electrode potentials of aluminum alloys and intermetallic compounds.
| Alloy or intermetallic compound | View semi-finished product | Stationary potential, mV |
| D16chT | panel | -595 |
| 1973T2 | plate | -685 |
| V95pchT2 | pressed strip | -675 |
| V95pchT3 | stamping | -700 |
| Al2Mg3Zn3 | — | -1215 |
| CuAl2 | — | -610 |
| Al2CuMg | — | -720 |
* The potential established after 24 hours in a 3% sodium chloride solution across a silver chloride electrode was taken as stationary potentials.
The addition of more electronegative components, such as magnesium, increases the passivation of aluminum, thereby increasing corrosion resistance.
The most popular coatings for protecting aluminum and its alloys from corrosion are anodizing, electroless nickel plating, and multilayer coatings based on copper, nickel, silver, and tin.
5.3 Corrosion of copper and its alloys.
Pure metallic copper has a potential of 0.337 V, due to which it has good corrosion resistance in normal atmosphere. It is not destroyed by non-oxidizing acids or in the presence of a depassivator. The destruction of copper begins in the presence of oxidizing acids (nitric acid, etc.), organic acids, and complexing agents.
Despite the fact that copper practically does not interact with oxygen, upon contact with humid air, copper begins to deteriorate. The resulting corrosion products are insoluble and prevent further corrosion of the metal.
2Cu + H2O + CO2 + O2 → CuCO3 + Cu(OH)2↓
Due to its high thermal conductivity and good corrosion resistance in water, copper is popular in heat exchanger or piping designs. However, copper is susceptible to cavitation and erosion corrosion. To prevent this corrosion, copper is alloyed.
In most cases, alloying elements give copper not only higher corrosion resistance, but also improve its mechanical properties. The most common alloying elements are zinc, nickel, tin, aluminum and silicon.
Alloys of copper and zinc, with the latter content ranging from 15% to 50%, are called brasses. Brass has high strength, good machinability and resistance to sulfides. Zinc, being a more reactive metal compared to copper, is easily subject to selective leaching. In this case, copper deposits of a characteristic red color can be observed on the surface of the brass.
Among other copper alloys, the alloy with tin is of particular importance. The addition of tin significantly increases the fatigue limit and at the same time allows maintaining corrosion resistance.
5.4 Corrosion of other metals.
Titanium
is a cathode metal relative to most other metals and alloys. An oxide film easily forms on it, resistant to most acids, including hydrochloric acid. Due to the combination of its properties: high strength, low specific gravity, high corrosion resistance, resistance to high temperature oxidation and high melting point, titanium is used in aircraft construction.
Nickel
has a number of physical and mechanical properties comparable to low-carbon steel. At the same time, nickel has higher corrosion resistance. As an alloying element, nickel increases the corrosion resistance of metals such as iron and copper. Pure nickel is almost not subject to contact corrosion, since, due to the formation of a protective oxide film, it is a cathode with respect to most metals. The nickel oxide film is resistant to alkalis.
Cobalt
in many respects similar to nickel. When combined with chromium, cobalt forms alloys that have excellent resistance to high temperature oxidation. Like nickel, cobalt can be easily passivated, especially when chromium is added to it.
Lead
demonstrates high corrosion resistance to sulfuric acid. Lead is used to cover containers and pipes that come into contact with this acid. The addition of 3-6% antimony increases the mechanical strength of lead. However, too much antimony is undesirable because it impairs the corrosive properties of lead; antimony is easily attacked by sulfuric acid.
Zinc
and its alloys cannot be used in corrosive atmospheres. This metal is too anodic and does not form virtually any protective film.
Almost all noble metals demonstrate high corrosion resistance in all acidic and alkaline environments. Silver and its alloys dissolve in oxidizing acids, but are resistant to reducing ones. Gold and platinum are resistant to even oxidizing acids. They are soluble in so-called aqua regia.
Methods for removing corrosion
If rust has already appeared, it can be removed in different ways - mechanically, chemically. You can also use folk remedies.
Rusty castle (Photo: pixabay.com)
Mechanical cleaning
Involves the use of abrasive tools. Damaged parts will be cleaned by friction.
Brush on metal
It is a classic hand brush with many metal fibers used for cleaning. Suitable for partial removal of the effects of corrosion.
Types of corrosion according to the nature of destruction:
- continuous (general corrosion) - covering the entire surface of the metal, which is under the influence of a corrosive environment;
- local - spreads only to certain areas of the metal surface.
Continuous corrosion is divided into: uniform, uneven and selective.
Local types of corrosion include: staining, pitting, pitting, through-hole, filamentous, intergranular, subsurface, knife, corrosion cracking and corrosion brittleness.
Recommendations
Adviсe:
- It is better not to skimp on protecting parts and cover them with rubber or polymer paint.
- Before using abrasives, you should try to remove rust with gentle compounds.
- Complex corrosion processes can be stopped with the help of aggressive chemicals, but before using them, you need to study the properties of the composition and the characteristics of the metal in order to prevent possible negative reactions.
Immediately after removing rust, surfaces should be coated with a protective compound to reduce the risk of corrosion re-spread.
Corrosive processes can quickly destroy any material. Damage to metal structures in some situations can have catastrophic consequences. Having studied the methods of protection against corrosion, you need to use one of the most suitable ones.
Why does metal corrode?
The life of a modern person cannot be imagined without metals. They surround us everywhere - these are household appliances in our homes, and the vehicles we use to get to home or work, and smartphones, without which many of us cannot imagine life. Almost everything that surrounds us consists of metals, but, unfortunately, like everything in this world, they are not eternal and under the influence of the external environment they are destroyed - they corrode.
Why is corrosion “beneficial” for metals? The fact is that most of them exist in nature in a chemically bound state, for example, in the form of oxides (corundum) or sulfides (pyrite). In their pure form, almost all metals are unstable and considerable energy must be expended to separate them from their compounds. The reverse process, when metals go into a bound state, is always thermodynamically more favorable. Therefore, it occurs spontaneously, and metals, whenever possible, tend to react with their environment and transform into a more stable form. An illustration of this is shown in Figure 1.
Figure 1 – Scheme of metal recovery from ores followed by corrosion (oxidation). E – conditional energy level. Corrosion leads to huge economic costs, and its consequences are global environmental disasters. The loss of metal stock from corrosion is about 12% per year.
In addition to direct losses, there are also indirect losses caused by corrosion:
- due to equipment downtime caused by accidents;
- due to reduced equipment capacity;
- due to a decrease in product quality;
- to eliminate the consequences of the accident;
- for equipment repair;
- for further protection against corrosion.
Methods to combat general corrosion
A method to prevent this type of corrosion is to evaluate the Corrosion Allowance (CA). This parameter determines the increase in metal thickness to predict material loss over the life of a part subject to corrosion. Since penetration depth may vary from case to case, the risk of corrosion is a safety factor.
The formula for calculating CA is:
CA = CR t
Knowing the expected corrosion rate (CR), expressed in µm/year, and the expected lifespan of mechanical parts (t), industries can calculate the additional thickness required for corrosion resistance of a material during the design process. The CA coefficient should be calculated according to the conditions of use of the metal piece.
The parameters that influence the corrosion rate (CR) are:
- Temperature;
- Humidity;
- Wind;
- Rain;
- Impurities;
- Aggressive substances.