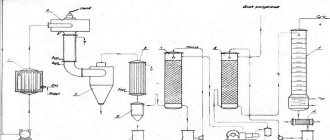
Copper from copper (II) sulfate can be obtained by the hydrometallurgical method:
At temperatures above 337 o C, copper reacts with oxygen to form copper (II) oxide:
Copper(II) oxide dissolves in dilute nitric acid to form copper(II) nitrate and water:
| Solution | Let's write down the reaction equations: |
Let's calculate the amount of copper (II) hydroxide substance (precipitate) (molar mass is 98 g/mol):
Let us determine the amount of substance and mass of copper (cathode) at the end of the reaction (molar mass – 64 g/mol):
Let's find the mass of copper deposited on the cathode:
Let us calculate the mass of the anode at the end of the reaction. The mass of the anode decreased exactly as much as the mass of the cathode increased:
Source
General characteristics of copper
The total content of copper in the earth's crust is relatively small [0.01% (wt.)], however, it is more often than other metals found in a native state, and copper nuggets reach a significant size.
The most important minerals that make up copper ores are: chalcocite, or copper luster Cu2S; chalcopyrite, or copper pyrite CuFeS2; malachite (CuOH)2CO3.
Pure copper is a viscous, viscous metal of light pink color (Fig. 1), easily rolled into thin sheets. It conducts heat and electricity very well, second only to silver in this regard. In dry air, copper remains almost unchanged, since the thin film of oxides that forms on its surface (giving copper a darker color) serves as good protection against further oxidation. But in the presence of moisture and carbon dioxide, the copper surface becomes covered with a greenish coating of hydroxycopper carbonate (CuOH)2CO3.
History of the discovery of Copper Cuprum
Discovery of the element Cuprum is one of the first metals to be well mastered by man due to its availability from ore and low melting point. This metal is found in nature in native form more often than gold, silver and iron. Some of the most ancient copper products, as well as slag - evidence of its smelting from ores - were found in Turkey, during excavations of the settlement of Çatalhöyük.
Copper Age
Copper objects became widespread, following the Stone Age in world history. Despite the softness of copper, copper tools, compared to stone ones, provide a significant advantage in the speed of cutting, planing, drilling and sawing wood, and processing bone takes approximately the same time as for stone tools.
In ancient times, copper was also used in the form of an alloy with tin - bronze - for the manufacture of weapons, etc., the Bronze Age replaced the Copper Age. An alloy of copper and tin (bronze) was produced for the first time in 3000 BC. e. in the Middle East. Bronze attracted people because of its strength and good malleability, which made it suitable for making labor and hunting tools, dishes, and jewelry. All these items are found in archaeological excavations. The Bronze Age regarding tools was replaced by the Iron Age.
Copper was originally mined from malachite ore rather than sulfide ore, as it does not require pre-roasting. To do this, a mixture of ore and coal was placed in a clay vessel, the vessel was placed in a small pit, and the mixture was set on fire. The released carbon monoxide reduced malachite to free copper:
In Cyprus, already in the 3rd millennium BC there were copper mines and copper smelting was carried out.
Copper mines appeared on the territory of Russia and neighboring countries two millennia BC. e. Their remains are found in the Urals (the most famous deposit is Kargaly), in Transcaucasia, in Siberia, Altai, and in Ukraine.
In the XIII-XIV centuries. mastered industrial copper smelting. In Moscow in the 15th century. The Cannon Yard was founded, where guns of various calibers were cast from bronze. A lot of copper was used to make bells. Such works of casting art as the Tsar Cannon (1586), the Tsar Bell (1735), the Bronze Horseman (1782) were cast from bronze, and a statue of the Big Buddha was cast in Japan (Todai-ji Temple) ( 752).
With the discovery of electricity in the 18th-19th centuries. large volumes of copper began to be used for the production of wires and other related products. And although in the 20th century. wires often began to be made of aluminum; copper has not lost its importance in electrical engineering.
Isotopes of copper
It is known that in nature copper can be found in the form of two stable isotopes 63 Cu (69.1%) and 65 Cu (30.9%). Their mass numbers are 63 and 65, respectively. The nucleus of an atom of the copper isotope 63 Cu contains twenty-nine protons and thirty-four neutrons, and the isotope 65 Cu contains the same number of protons and thirty-six neutrons.
There are artificial unstable isotopes of copper with mass numbers from 52 to 80, as well as seven isomeric states of nuclei, among which the longest-lived isotope 67 Cu with a half-life of 62 hours.
Molar mass of copper:
Molar mass is a characteristic of a substance, the ratio of the mass of a substance to its quantity.
In the International System of Units (SI), the unit of measurement for molar mass is kilogram per mole (Russian designation: kg/mol; international: kg/mol). Historically, molar mass was typically expressed in g/mol.
The molar mass is numerically equal to the mass of one mole of a substance, that is, the mass of a substance containing a number of particles equal to Avogadro’s number (NA = 6.022 140 76⋅10 23 mol −1).
Molar mass, expressed in g/mol, is numerically the same as molecular mass (absolute molecular mass), expressed in a. e.m., and relative molecular weight.
In turn, molecular mass is the mass of a molecule. There are absolute molecular mass (usually expressed in atomic mass units, amu) and relative molecular mass - a dimensionless value equal to the ratio of the mass of a molecule to 1/12 of the mass of a 12 C carbon atom.
Molar mass is denoted by M.
The molar mass of copper (M (Cu)) is 63.546(3) g/mol.
It must be borne in mind that the molar masses of chemical elements and the simple substances that they form are not the same thing. For example, the molar mass of oxygen as a chemical element (atom) is ≈ 16 g/mol, and the substance (O2) is ≈ 32 g/mol.
Source
Copper Cuprum origin of the name
Where does the name Cuprum come from? The Latin name for copper Cuprum (ancient Aes cuprium, Aes cyprium) comes from the name of the island of Cyprus, where there was a rich deposit.
Strabo calls copper chalkos, from the name of the city of Chalkis on Euboea. From this word came many ancient Greek names for copper and bronze objects, blacksmithing, blacksmithing and casting. The second Latin name for copper Aes (Sanskrit ayas, Gothic aiz, German erz, English ore) means ore or mine.
The words copper and copper are found in the most ancient Russian literary monuments. Slavic *mědь “copper” does not have a clear etymology, perhaps an original word. V.I. Abaev assumed the origin of the word from the name of the country Media: *Copper from Ir. Māda - through the Greek. Μηδία[8]. According to the etymology of M. Vasmer, the word “copper” is related to ancient German. smid “blacksmith”, smîda “metal”.
Copper was designated by the alchemical symbol “♀” - “mirror of Venus”, and sometimes copper itself was also called “Venus” by alchemists. This is due to the fact that the goddess of beauty, Venus (Aphrodite), was the goddess of Cyprus, and mirrors were made from copper. This symbol of Venus was also depicted on the brand of the Polevsky copper smelter, it was used to brand Polevsky copper from 1735 to 1759, and is depicted on the modern coat of arms of the city of Polevskoy. Polevsky’s Gumeshevsky mine, the largest copper ore deposit of the Russian Empire in the Middle Urals in the 18th-19th centuries, is associated with a famous character in P. P. Bazhov’s fairy tales - the Mistress of the Copper Mountain, the patroness of the mining of malachite and copper. According to one hypothesis, she is the image of the goddess Venus refracted by the popular consciousness[
Prevalence of Copper Cuprum
Like any chemical element has its abundance in nature, Cu...
Brief theory
Molar mass
(Μ or μ) - the mass of one mole of a substance in grams, i.e. how much N = 6.02×10 23 molecules or other particles weigh (N = NA - Avogadro's number). Example entry: M(H2O) = 18 g/mol
(Relative) molecular weight
(Mr - for molecules and Ar - for atoms) - the mass of one molecule or other particle in atomic mass units (amu) or daltons (Da). Example notation: Mr(H2O) = 2×Ar(H) + Ar(O) = 2×1 + 16 = 18 (a.u.m)
1 amu = mu = 1/12 mass of carbon nuclide 12 C = 1.660×10 -24 g, where mu is the unified mass. In this case, mu×NA ≡ 10 -3 Molar and molecular masses are numerically equal, but have different meanings (and dimensions). The mass of one molecule (in grams) is found as the product: mu×Mr = m(molecules). Then the connection of all three masses
between themselves: M = m(molecules)×NA = mu×Mr×NA.
Instructions for using the calculator
Main characteristics of the calculator:
- Understands elements from H to Uuh
- Supports arbitrary nesting of ligands (square and parentheses) - for example, the “hypothetical” substance Li2[(H2O)2O(CH4[NH3]2)]3[CoSeMg]2
- Inclusions (such as crystalline hydrates) can be designated by: * × ∙ · ⋅ ∗
The calculator does not yet support:
- Abbreviations such as Ph, Met, Ac and the like
- Settable accuracy of mass calculation (to an arbitrary decimal place)
Source
1. What area can a drop of olive oil with a volume of 0.02 cm3 occupy when it spreads on the surface of water?
2. Determine the molar masses of hydrogen and helium.
3. How many times is the number of atoms in carbon weighing 12 kg greater than the number of molecules in oxygen weighing 16 kg?
4. What is the amount of substance (in moles) contained in water weighing 1 g?
5. The molar mass of nitrogen is 0.028 kg/mol. What is the mass of a nitrogen molecule?
6. Determine the number of atoms in copper with a volume of 1 m3. The molar mass of copper is M = 0.0635 kg/mol, its density is ρ = 9000 kg/m3.
7. Diamond density 3500 kg/m3. What volume will 1022 atoms of this substance occupy?
8. At what pressure is the gas in the vessel if the average square of the speed of its molecules is v2 = 106 m2/s2, the concentration of molecules is n = 3 • 1025 m-3, the mass of each molecule is m0 = 5 • 10-26 kg?
9. A 1.2 liter flask contains 3 •1022 helium atoms. What is the average kinetic energy of each atom? The gas pressure in the flask is 105 Pa.
10. Calculate the average square of the speed of movement of gas molecules if its mass is m = 6 kg, volume V = 4.9 m3 and pressure p = 200 kPa.
Source
Relative density of one gas to another
Sometimes, to solve a problem, you need to know how to find the molar mass of a gas, for which only its density in air or another gas is reported. This is possible if you know the relative density formula, which is denoted by the letter D.
, where and are some gases.
Example 1
How to determine how many times carbon monoxide is denser than hydrogen?
First, let's find the molar mass of CO and H2:
M(CO) = Mr(CO) = Ar(C) + Ar(O) = 12 + 16 = 28.
M(H) = Mr(H2) = 2 Ar(H) = 2 1 = 2.
.
Answer: Carbon monoxide is 14 times denser than hydrogen.
Example 2
How to calculate the molar mass of a gas x, which is known to be 10 times denser than carbon dioxide CO2?
First, let's calculate the molar mass of carbon dioxide:
M(CO2) = Mr(CO2) = 12 + 2 16 = 44.
Based on the relative density formula, we will calculate the molar mass of the desired gas x.
M(x) = D(x/CO2) M(CO2) = 10 44 = 440 g/mol.
Answer: This gas has a molar mass of 440 g/mol.
Copper atom and molecule. Copper formula. Structure of the copper atom:
Copper (lat. Cuprum) is a chemical element of the periodic system of chemical elements of D.I. Mendeleev with the designation Cu and atomic number 29. It is located in the 11th group (according to the old classification - a secondary subgroup of the first group), the fourth period of the periodic system.
Copper is a metal. Belongs to the group of transition metals. Refers to heavy and non-ferrous metals.
As a simple substance , copper under normal conditions is a ductile metal of golden-pink color (or pink in the absence of an oxide film). Along with osmium, cesium and gold, copper is one of the four metals that have a distinct coloration that is different from the gray or silver of other metals.
The copper molecule is monatomic.
The chemical formula of copper is Cu.
The electronic configuration of the copper atom is 1s 2 2s 2 2p 6 3s 2 3p 6 3d 10 4s 1. The ionization potential (first electron) of the copper atom is 745.48 kJ/mol (7.726380(4) eV).
Structure of the copper atom. The copper atom consists of a positively charged nucleus (+29), around which 29 electrons move in four shells. In this case, 28 electrons are in the internal level, and 1 electron is in the external level. Since copper is located in the fourth period, there are only four shells. First, the inner shell is represented by the s-orbital. The second – the inner shell is represented by s- and p-orbitals. The third - inner shell is represented by s-, p- and d-orbitals. The fourth - outer shell is represented by the s-orbital. At the outer energy level of the copper atom, the s orbital contains one unpaired electron. In turn, the nucleus of a copper atom consists of 29 protons and 35 neutrons.
The radius of the copper atom (calculated) is 145 pm.
The atomic mass copper atom is 63.546(3) a. eat.
Copper has been widely used by humans for a long time.
Zinc, Zn
Latin name Zincum, chemical symbol Zn. Element of period 4, located in group II, B-subgroup. Serial number 30. Mass - 65.37. Structure of electronic shells: 1s2 2s22p6 3s23p63d10 4s2 (in the ground state). Valency and oxidation state: II(+) and +2 (respectively).
Methods of production in industry:
- Reduction by carbon upon heating: ZnO+ C→ CO↑ + Zn.
- Hydrometallurgy: ZnO + H2SO4 → ZnSO4+ H2O; ZnSO4+ Fe → FeSO4+ Zn↓.
- Electrolysis: zinc is reduced at the cathode Zn2+ + 2H+ + 4ē → Zn↓ + H2.
Zinc is a silver-gray metal ( Fig. 3 ). Solid, conducts heat and electricity. Oxidized by oxygen when heated. Does not interact with boron, carbon, silicon, nitrogen. It does not dissolve in water, but upon strong heating it reacts with water vapor to form zinc oxide and release hydrogen. Reacts with acids other than nitric acid, displacing hydrogen. Displaces metals located to the right in the activity series from solutions of their salts.
Rice. 3. Zinc
table 2
Characteristics of connections
| Substance classes | Names and formulas | Properties |
| Oxides | Zinc oxide, ZnO | Amphoteric. |
| Hydroxides | Zinc hydroxide Zn(OH)2 | Amphoteric. |
Zinc is used as a protective material to prevent rust (galvanizing) of steel and iron products. The metal is used in construction, the production of household appliances and for other purposes.