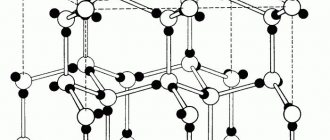
Graphite (from ancient Greek γράφω - I write) is a natural material belonging to the class of native elements, an allotropic modification of carbon. It has a layered structure. Each layer of the graphite crystal lattice can be positioned differently in relation to each other, forming polytypes.
Graphite finds its application in manufacturing and industrial activities. Graphite products are distinguished by increased performance characteristics. Graphite is resistant to chemical and natural influences, it is quite durable, conducts electricity well, has low hardness, relative softness, and hardens after exposure to high temperatures.
The density is 2.23 g/cm3. Graphite has a metallic luster and dark gray color. The thermal conductivity of this mineral is quite high, so it is used for the manufacture of components for electrical equipment.
There are deposits of crystalline graphite associated with igneous rocks or crystalline shales, and cryptocrystalline graphite formed during the metamorphism of coals.
Graphite structure
Hexagonal crystalline polymorphic (allotropic) modification of pure carbon, the most stable under the conditions of the earth's crust.
The layers of the crystal lattice can be differently positioned relative to each other, forming a whole range of polytypes, with symmetry from hexagonal system (dihexagonal-dipyramidal type of symmetry) to trigonal (ditrigonal-scalenohedral symmetry). The crystal lattice of graphite is layered.
In layers, C atoms are located at the sites of hexagonal cells of the layer. Each C atom is surrounded by three neighboring ones with a distance of 1.42Α
There are two modifications of graphite: α-graphite (hexagonal P63/mmc) and β-graphite (rhombohedral R(-3)m). They differ in the packing of layers. In α-graphite, half of the atoms of each layer are located above and below the centers of the hexagon (laying...AVAVAVA...), and in β-graphite, every fourth layer repeats the first. It is convenient to represent rhombohedral graphite along hexagonal axes to show its layered structure.
β-graphite is not observed in its pure form, since it is a metastable phase. However, in natural graphites the content of the rhombohedral phase can reach 30%. At a temperature of 2500-3300 K, rhombohedral graphite completely transforms into hexagonal graphite.
Crystal lattices
The crystal lattice is the spatial arrangement of atoms or ions in a crystal. The points of the crystal lattice at which atoms or ions are located are called lattice nodes.
Crystal lattices are divided into molecular, atomic, ionic and metallic.
It is very important not to confuse the type of chemical bond and crystal lattice. Remember that crystal lattices reflect the spatial arrangement of atoms.
Molecular crystal lattice
Molecules are located at the nodes of a molecular lattice. Under normal conditions, most gases and liquids have a molecular lattice. Bonds are most often covalent polar or nonpolar.
The classic example of a substance with a molecular lattice is water, so associate the properties of these substances with water. Substances with a molecular lattice are fragile, have low hardness, are volatile, fusible, capable of sublimation, and are characterized by low boiling points.
Examples: NH3, H2O, Cl2, CO2, N2, Br2, H2, I2. I would especially like to mention white phosphorus, orthorhombic, plastic and monoclinic sulfur, and fullerene. We studied these allotropic modifications in detail in an article devoted to the classification of substances.
Ionic crystal lattice
At the sites of the ionic lattice there are atoms connected by ionic bonds. This type of lattice is characteristic of substances that have ionic bonds: metal salts, oxides and hydroxides.
Metal crystal lattice
The nodes of the metal lattice contain metal atoms. This type of lattice is characteristic of substances formed by a metallic bond.
Associate the properties of these substances with copper. They have a characteristic metallic luster, are malleable and ductile, conduct electricity and heat well, and have high melting and boiling points.
Examples: Cu, Fe, Zn, Al, Cr, Mn.
Atomic crystal lattice
At the sites of the atomic lattice there are atoms connected by covalent polar or nonpolar bonds.
Associate these substances with sand. They are very hard, very refractory (high melting point), non-volatile, durable, and insoluble in water.
Examples: SiO2, B, Ge, SiC, Al2O3. I would especially like to highlight: diamond and graphite (C), red and black phosphorus (P).
© Bellevich Yuri Sergeevich 2018-2021
This article was written by Yuri Sergeevich Bellevich and is his intellectual property. Copying, distribution (including by copying to other sites and resources on the Internet) or any other use of information and objects without the prior consent of the copyright holder is punishable by law. To obtain article materials and permission to use them, please contact Yuri Bellevich
.
Source
Properties
Conducts electricity well. Unlike diamond, it has low hardness (1 on the Mohs scale). Relatively soft. After exposure to high temperatures it becomes slightly harder and becomes very brittle. Density 2.08-2.23 g/cm³.
The color is dark gray, metallic luster. Infusible, stable when heated in the absence of air. Greasy (slippery) to the touch. Natural graphite contains 10-12% admixtures of clays and iron oxides. When rubbed, it separates into separate flakes (this property is used in pencils).
The thermal conductivity of graphite is from 278.4 to 2435 W/(m*K), depending on the grade of graphite, on the direction relative to the basal planes and on temperature.
The electrical conductivity of graphite single crystals is anisotropic, in the direction parallel to the basal plane it is close to the metallic one, in the perpendicular direction it is hundreds of times less.
The minimum conductivity value is observed in the range of 300–1300 K, and the position of the minimum shifts to the region of low temperatures for perfect crystalline structures. Recrystallized graphite has the highest electrical conductivity.
The coefficient of thermal expansion of graphite up to 700 K is negative in the direction of the basal planes (graphite contracts when heated), its absolute value decreases with increasing temperature. Above 700 K the coefficient of thermal expansion becomes positive.
In the direction perpendicular to the basal planes, the coefficient of thermal expansion is positive, practically independent of temperature and more than 20 times higher than the average absolute value for the basal planes.
Graphite single crystals are diamagnetic; the magnetic susceptibility is insignificant in the basal plane and high in planes orthogonal to the basal planes. The Hall coefficient changes from positive to negative at 2400 K.
Graphite has a crystal lattice
The table below lists the characteristic properties of substances with atomic and ionic crystal lattices.
Characteristic properties of substances
- solid under normal conditions;
- conduct electric current in melts and solutions
| With atomic crystal lattice | With ionic crystal lattice |
Using this information, determine what kind of crystal lattice it has:
1) calcium chloride
;
2) graphite
.
Write your answer in the space provided:
1) Calcium chloride has
Calcium chloride is a substance with an ionic chemical bond, refractory (T mp = 772 °C), conducts electric current - has an ionic crystal lattice.
Graphite is a substance with a covalent nonpolar chemical bond, nonvolatile, solid, and has an atomic crystal lattice.
Answer: Calcium chloride is an ionic crystal lattice, graphite is an atomic crystal lattice.
Source
Morphology
Well-formed crystals are rare. The crystals are lamellar, scaly, curved, and usually have an imperfect lamellar shape. More often it is represented by leaves without crystallographic outlines and their aggregates.
Forms continuous cryptocrystalline, foliated or round radial-radiating aggregates, less often - spherulitic aggregates of a concentric-zonal structure. Coarse-crystalline precipitates often exhibit triangular shading on the (0001) planes.
Story
The history of the mineral graphite is quite confusing and difficult to study, since the mineral is very similar to other stones. However, mineralogists studying the history of stones still find some information about the history of the origin and use of graphite.
They believe that graphite stone began to be used in medieval times. Wherever the stone was found on the surface, it left clear greasy stains. When ancient people noticed this, they began to use the stone for writing and drawing. Later, thanks to this property, the stone began to be used in England. Using graphite crayons, English shepherds made marks on the wool of sheep so as not to lose sight of them.
The very first artists created crayons and similarities to modern pencils from graphite. They used them to paint graffiti-style paintings. Engineers and architects used graphite fragments to make drawings when designing various buildings, structures, and temples.
Some researchers have found the first mention of graphite 4000 BC. They claim that the mineral was used by the primitive people of the Boyan Morica culture to decorate pottery. Using graphite stone, they painted household items in grayish and black colors, and also painted various patterns on them.
The history of the name of the stone is associated with the famous mineralogist and chemist Abram Werner. In the process of compiling a physical and chemical description of graphite, the scientist took as a basis the property of the stone to leave a black color behind itself. This explains the ancient Greek name for graphite, meaning “to write, record.”
An interesting fact is that the name of the discoverer of carbon is still unknown to science, as is the fact itself, which of its forms - diamond, graphite - was discovered first. It is only known that carbon has 3 isotopes, two of which are stable, and one is radioactive and has a half-life of more than 5 thousand years. A study of various 18th century chemistry textbooks also does not provide a clear answer about carbon derivatives. Among the experiments of ancient chemists, combustion of various chemical elements and allotropic substances was often used. Thus, in one of the works there is a mention of the burning of diamond, where it was emphasized that this mineral is the only one that burns without a residue. The scientist Lavoisier, conducting similar experiments, came to the conclusion that diamond is a crystalline form of carbon. The second allotropic mineral of carbon (graphite) was long considered a modified form of lead and was called plumbago, until the chemist Guiton de Morveau carefully heated diamond, turning it into graphite, and then into carbonic acid.
Origin
Formed at high temperatures in volcanic and igneous rocks, pegmatites and skarns. It is found in quartz veins with wolframite and other minerals in medium-temperature hydrothermal polymetallic deposits.
Widely distributed in metamorphic rocks - crystalline schists, gneisses, marbles. Large deposits are formed as a result of pyrolysis of coal under the influence of traps on coal deposits (Tunguska basin). Accessory mineral of meteorites. Associated minerals: quartz, pyrite, garnets, spinel.
Metal type
Its structure resembles a molecular one, but it still has stronger bonds. The difference between this type is that its nodes contain positively charged cations. Electrons that are located in the interstitial space participate in the formation of the electric field.
They are also called electric gas. Simple metals and alloys are characterized by a metal lattice type. They are characterized by the presence of a metallic luster, plasticity, thermal and electrical conductivity. They can melt at different temperatures.
Source
Application
For the manufacture of melting crucibles, lining plates - the application is based on the high temperature resistance of graphite (in the absence of oxygen), on its chemical resistance to a number of molten metals.
It is used in electrodes and heating elements due to its high electrical conductivity and chemical resistance to almost any aggressive aqueous solutions (much higher than that of noble metals).
For the production of chemically active metals by electrolysis of molten compounds, solid lubricants, in combined liquid and paste lubricants, plastic filler.
It is a neutron moderator in nuclear reactors, a component of the composition for the manufacture of leads for black graphite pencils (mixed with kaolin).
It is used to obtain synthetic diamonds, as a nanometer length standard for calibrating scanners of a scanning tunneling microscope and atomic force microscope, for the manufacture of contact brushes and current collectors for a variety of electrical machines, electric vehicles and overhead cranes with trolley power, powerful rheostats.
As well as other devices where reliable moving electrical contact is required, for the manufacture of thermal protection for the nose part of ballistic missile warheads and re-entry spacecraft.
Graphite - C
| Molecular weight | 12.01 g/mol |
| origin of name | from ancient Greek γράφω - to record, to write |
| IMA status | valid, first described before 1959 (before IMA) |
What are pearlite and eutectoid
Observations show that this transition occurs as follows: upon reaching temperatures GS along the boundaries Observations show that this transition occurs as follows: upon reaching temperatures GS along the boundaries of austenite crystals, the first portions of α - Fe, i.e., ferrite, are released, the amount of which gradually increases.
Since ferrite almost does not dissolve carbon, during the transition γ-Fe -> α-Fe, the carbon concentration in retained austenite gradually increases and can be determined by the GS line depending on temperature. The ferrite separation process continues until the carbon concentration corresponds to point 5, i.e., up to C = 0.83%, and the temperature reaches t = 723°.
At point S, the GS curve intersects with ES, the curve of the limiting solubility of carbon in austenite. Therefore, further saturation of retained austenite with carbon becomes impossible, and subsequent cooling causes the final decomposition of austenite, which occurs at a constant temperature t = 723°.
During this decomposition, the γ-Fe->α-Fe transition is completed, and the carbon released from the iron crystal lattice forms F3C cementite particles. The decomposition of austenite occurs in a confined space within each grain, so the decomposition products (ferrite and cementite) are formed in the form of closely mixed particles, usually in the form of alternating plates of ferrite and cementite.
Scheme of changes in the structure of steels when passing through critical points
This decomposition product of austenite is called pearlite; Since pearlite has a structure similar to eutectic, it is called a eutectoid. The difference between eutectic and eutectoid is that eutectic is formed from a liquid solution, while eutectoid is formed from a solid.
The formation of pearlite begins and ends at a constant t=723°. This is how the ferrite-pearlite structure of steels appears, which, upon further cooling from t = 723°, no longer undergoes any structural changes. The figure shows the microstructures of pure iron and steel at C = 0.15% and at C = 0.6% (magnification 100) after etching the scratched surface of a microsection with a 4% solution of HNO3 in ethyl alcohol.
Rice. 1. - ferrite in pure iron. Rice. 2 Hypoeutectoid steel with C content = 0.15%
In Fig. 1, which shows the microstructure of pure iron, the boundaries between the light ferrite grains are clearly visible. In Fig. Figure 2 shows the microstructure of building steel (C=0.15%); light fields are ferrite, dark areas are pearlite. In Fig. Figure 3 shows the microstructure of engineering steel (C = 0.6%), from which axles, shafts, connecting rods, etc. are made; Most of the section is occupied by pearlite, and ferrite is observed only in the form of a thin mesh. The more carbon, the more pearlite in the steel structure; the composition of pearlite is the same (C = 0.83%). The structure of perlite is usually lamellar (Fig. 4).
Rice. 3 Hypoeutectoid steel with C content = 0.6%. Rice. 4 Eutectoid steel (lamellar pearlite).
Ferrite, as mentioned above, is the softest plastic component of iron-carbon alloys; cementite, which is part of pearlite, is the hardest and most brittle, therefore, with increasing carbon content, the strength and hardness of steel increases, but ductility and toughness decrease
In order for construction steel to be sufficiently ductile, the amount of perlite in it should not exceed 25%, which corresponds to a carbon content of up to 0.2%.
In those parts that require greater strength and hardness, but lower ductility and toughness are acceptable (machine parts), steels with a large amount of pearlite, with a C content of up to 0.6%, are used. In the construction industry, such steels are used, for example, for the manufacture of shovels and supporting parts of bridge trusses.
Types of natural graphite
- crucible (used for the production of refractory products. It is characterized by increased thermal conductivity and resistance to sudden temperature changes),
- casting crystal (has a low coefficient of expansion, is characterized by strength at high temperatures, is used when casting parts),
- battery (used as an additive, graphite is used for the production of electrodes, has improved technical and chemical properties),
- for the production of pencil leads (fine, soft, does not contain iron impurities),
- elemental (graphite is used for the production of galvanic cells, it has increased thermal and electrical conductivity),
- electric coal,
- for the production of lubricants and electrically conductive rubber.
Definition
A crystal lattice is a special structure of a solid substance in which the particles stand in a geometrically strict order in space.
In it you can find nodes - places where elements are located: atoms, ions and molecules and internodal space. Solids , depending on the range of high and low temperatures, are crystalline or amorphous - they are characterized by the absence of a certain melting point. When exposed to elevated temperatures, they soften and gradually turn into liquid form. These types of substances include: resin, plasticine.
In this regard, it can be divided into several types:
But at different temperatures, one substance can have different forms and exhibit diverse properties. This phenomenon is called allotropic modification.
This is interesting: metals and non-metals in the periodic table of Mendeleev.
Physical properties
| Mineral color | iron black fading to steel gray |
| Stroke color | black fading to steel gray |
| Transparency | opaque |
| Shine | semi-metallic |
| Cleavage | very perfect in {0001} |
| Hardness (Mohs scale) | 1-2 |
| Kink | mica-like |
| Strength | flexible |
| Density (measured) | 2.09 – 2.23 g/cm3 |
| Radioactivity (GRapi) | 0 |
Graphite
Graphite
is a mineral from the class of native elements, one of the allotropic modifications of carbon. A common mineral in nature. It is usually found in the form of individual flakes, plates and clusters, varying in size and graphite content. There are deposits of crystalline graphite associated with igneous rocks or crystalline shales, and cryptocrystalline graphite formed during the metamorphism of coals.
Therapeutic effect
Graphite has a large number of healing properties, which allows folk medicine to actively use it as a remedy for diseases.
- Graphite has a beneficial effect on the layers of the epidermis. It is used in the treatment of skin cracks, scars, bruises, and eczema.
- Lithotherapists use graphite products to treat diseases in the sinus area. The stone helps get rid of dryness in the nasal mucosa, eliminate various chronic diseases of the respiratory tract, rhinitis, laryngotracheitis. Graphite is also used as a prophylactic treatment for bronchial asthma.
- Graphite stones help improve the condition of the gastrointestinal tract, improve metabolic processes in the body, fight chronic gastritis, eliminate heartburn and reduce the worsening of constipation or diarrhea.
- Healing graphite helps cope with severe headaches, apathy and stress.
- Graphite has a healing effect on human eyes. Small stones relieve inflammation in barley, conjunctivitis, and also help in the treatment of cataracts or corneal ulcers.
- The mineral regulates emotional balance, reducing the level of anger, temper, aggression, depression and neurasthenia.
Where is it used?
The use of graphite is an integral part of some technological processes. Used in industries such as mechanical engineering, nuclear engineering, metallurgy, electrical engineering, and the chemical industry.
graphite electrodes
graphite bushings
When creating chemical equipment, certain varieties of graphite impregnated with various synthetic resins are used. This is caused by the property of graphite not to react with oxidizing agents.
graphite sealant for pumps
Mechanical seals, bearings, reactor vessels, and lining tiles are made from artificial graphite.
How are graphite rods produced?
The production process is automated and divided into several stages. First, the lead itself is produced from a mixture of clay and graphite. The hardness of the future pencil depends on the ratio of the proportions of these materials. The more graphite, the softer the pencil.
The clay is crushed and mixed with warm water, then liquid glass is added to remove impurities (sand). Next, graphite is added to the clay according to the recipe. This whole mass is mixed with a starch binder.
The core mass is brought to a certain temperature and consistency. At this stage of production, strict process rules must be followed, otherwise the raw materials will be spoiled. The resulting “dough” is passed through a press, in which the required product with three gaps is formed through special rollers. This process removes air bubbles. If this stage occurs in violation of technology, then the future pencil will crumble when pressed.
Next, the mass takes on the desired shape and is glued between two planks. The result is a familiar pencil to all of us.
Place of Birth
Graphite mine
The demand for graphite in industry is very high. Today, approximate reserves around the world are estimated at 600 million tons. The largest graphite deposits are located in China, Mexico, Russia, the Czech Republic, South Korea and other countries. In addition to the countries listed, graphite is also mined on the island of Sri Lanka. Large reserves of this mineral were also found in Ukraine, in the so-called Zavalevskoe deposit. The discovered graphite deposits are industrially significant and are in great demand.
Where and how is it mined?
Graphite is mined all over the world. The ranking of countries by reserves and production in the world is presented below.
- China . About 780 thousand tons per year. In 2022, the country became the largest producer of this mineral in the world. During the year, China produced the same amount of graphite as in the previous 2 years. The US Geological Survey estimates that the country accounts for 65% of global production and 35% of consumption.
- India. 150 thousand tons were produced. Deposit reserves vary from state to state. 43% of the reserves are in Arunachal Pradesh. In total, there are 8 graphite mining and production companies in the country.
- Brazil . Production is 90 thousand tons per year. All leading manufacturing companies are private. The production level remains the same, but there are prerequisites for companies from China and India to squeeze them out of the market.
- Canada . 30 thousand tons of graphite are mined per year. Demand for Canadian graphite is growing, especially after Tesla announced it would purchase graphite for battery production from companies based in Canada.
- Mozambique . Production 23 thousand tons. Graphite mining in this country has grown from zero in 2016 to present in 2022. The pace of production is constantly increasing, the market in this region is represented by two companies.
Other countries produce less than 20 thousand tons per year. The top ten countries are rounded out by Russia (6th place), Ukraine, Pakistan, Norway and Madagascar. In total, these countries mine about 63 thousand tons of graphite per year. The graphite market in the world is constantly growing. This is due to the transition to electric batteries in many industries. The surge in electric vehicle production around the world is stimulating demand for graphite.
World consumption grew from 2013 to 2022, then there was a decrease in demand for this mineral. Prices dropped to 2017 levels as there was an oversupply and companies reduced production to stabilize world prices for the mineral.